By Bahram Robert Oliai, M.D.
In 1907 Dr. George Hoyt Whipple of The Johns Hopkins Hospital (left) performed an autopsy on a 36-year-old medical missionary, who succumbed to a strange illness. Review of tissue samples from the case demonstrated a peculiar accumulation of foamy macrophages within both the lymph nodes and in the lamina propria of the small bowel, findings characteristic of the disease that would eventually come to bear his name. Although initially named “intestinal lipodystrophy,” in the 1990s, the causative organism of Whipple’s disease was specifically identified as Tropheryma whipplei (an acinetobacterium) using PCR technology.
In this month’s Focus on Immunohistochemistry, we review this fascinating disease and discuss a new, extremely useful, and quite a specific method of making the diagnosis of Whipple’s disease now available to ProPath clients!
Whipple’s Disease
Whipple’s disease is a rare multisystem infectious disorder, which tends to more commonly affect middle-aged Caucasian men, although current trends show increasing infection rates among females and in older patients. While not universally accepted, there is some data to suggest that the disease is more common among farmers and carpenters, and host factors may be involved. Specifically, there are some reports implicating an increased susceptibility in those with histocompatibility antigen HLA-B27.
The clinical features of Whipple disease are protean. While the characteristic clinical picture is that of weight loss, chronic diarrhea, arthralgia, and fever with lymphadenopathy (sometimes so extreme as to cause tumoral masses), patients can also manifest anemia, hepatosplenomegaly, cardiovascular dysfunction, pulmonary infiltrates, and central nervous system/ocular dysfunction.
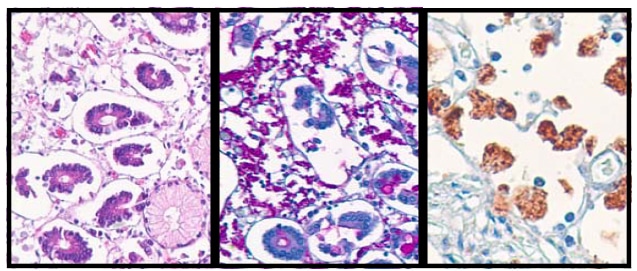
Whipple’s Disease of the small bowel, stained with H&E (left), digested PAS (center) and immunostained with the Whipple’s Disease antibody (right). The immunostain selectively and specifically stains the bacteria-filled histiocytes.
Diagnostic Modalities
Unfortunately, there are currently no specific laboratory abnormalities which can be used to make the diagnosis of Whipple’s disease. Culture is often not feasible as this is a rather fastidious organism, and in cases in which patients have already started antibiotic therapy, the recovery can be very poor. Although serological tests are available and IgM titers above 1:400 have shown some degree of specificity for Whipple’s disease, serologic studies are plagued by cross-reactivity with other microbes and the ubiquitous distribution of both virulent and nonvirulent strains of T. whipplei.
The relationship of Whipple’s Disease with The Johns Hopkins Hospital continued, and in 1961, Drs. Yardley and Hendrix described the characteristic electron microscopic finding of a trilaminar bacterial cell wall both within macrophages and extracellularly. They termed these structures “bacillary bones” and for many years EM became the mainstay of diagnosis. While quite specific, EM is expensive (now often only available at large research institutions), time-consuming, and turn around time is often poor.
Polymerase chain reaction tests may also be used and generally exhibit good sensitivity. However, a positive PCR test is not always specific for the disease, as T. whipplei DNA has been recovered from healthy patients. Due to variable specificity, one may need to perform PCR tests using primers from two genes to avoid false positives. This adds to the expense of the test, and because of this many PCR tests need to be “batched,” increasing the length of turn around time.
Currently, most cases of Whipple’s disease are diagnosed on duodenal biopsy. The classically described, endoscopic finding is of thickened mucosal folds and multiple whitish-yellow plaques on the intestinal mucosa. The classic histologic finding, described by a Whipple almost 100 years ago, is the presence of foamy, vacuolated macrophages, which infiltrate the lamina propria and stain PAS positive. The intestinal villi are often thickened and somewhat blunted. These foamy PAS-positive macrophages are actually histiocytes that are “busting at their seams” with the causative rod-shaped bacteria. Although they are a characteristic finding in Whipple’s disease, PAS-positive foamy macrophages are not entirely specific and similar macrophages can be seen in infection with Rhodococcus equi, Histoplasma capsulatum, Bacillus cereus, and Mycobacterium avium-intracellulare. In addition, PAS-positive macrophages can be seen in intestinal histiocytosis, sarcoidosis, Gaucher’s disease, and berylliosis.
Why Immunohistochemistry?
Once the Whipple’s Bacillus had been cultivated, a group of physicians from Marseille France developed a highly specific and sensitive polyclonal rabbit anti-T. whipplei immunohistochemical stain. This immunoperoxidase stain can easily be performed on formalin-fixed paraffin-embedded biopsy tissue in a timely and cost-effective fashion, allowing for a specific diagnosis without waiting for culture, EM, or PCR. This antibody has shown itself to be quite robust. In fact a friend of mine from residency, Dr. Blaire Baisden, one of my former attendings Dr. Stephen Dumler, and Dr. Didier Raoult (of the group from Marseille that raised the antibody) were able to retrieve tissue from the original autopsy case described by Dr. Whipple and demonstrate positive staining within the small bowel tissue nearly 100 years later!
At ProPath we now have this remarkable antibody and have titered it successfully within our laboratory. We are ready to offer this highly sensitive and specific immunohistochemical stain which can be performed on formalin-fixed paraffin-embedded tissue (even from remote cases) often with a turn around time of less than 24 hours.
Acknowledgments:
I would like to thank Drs. Florence Fenollar and Didier Raoult of the Unite des Rickettsies, Marseilles France for their kindness and generosity in allowing us the use of their antibody and Dr. Grover Hutchins of The Johns Hopkins Hospital for all his help in helping us procure control tissue.
References:
1. Dumler JS, Baisden BL, Yardley JH, Raoult D. Immunodetection of Tropheryma whipplei in Intestinal Tissues from Dr. Whipple’s 1907 Patient. New England Journal of Medicine 2003; 348(14): 1411-1412.
2. Bai JC, Mazure RM, Vazquez H, Niveloni SI, Edgardo S, Pedreira S, Maurino E. Whipple’s Disease. Clinical Gastroenterology and Hepatology 2004; 2(10): 849-860.
3. Lepidi H, Fenollar F, Gerolami R, Mege JL, Bonzi MF, Chappuis M, Sahel J, Raoult D. Whipple’s Disease: Immunospecific and Quantitative Immunohistochemical Study of Intestinal Biopsy Specimens. Human Pathology 2003; 34(6): 589-596.
4. Marth T, Raoult D. Whipple’s Disease. The Lancet 2003; 361(9353): 239-246.
5. Muir-Padilla J, Myers J. Whipple Disease A Case Report and Review of the Literature. Archives of Pathology and Laboratory Medicine. 2005; 129: 933-936.
